How the spray works. Image – University of Sydney, University of Melbourne.
A SPRAYABLE coating that prevents the surface spread of infection from bacteria and viruses, including COVID-19, could also have a role in Foot and Mouth Disease risk management, according to one of the lead researchers.
The first-of-its-kind sprayable coating can prevent the surface spread of infection from bacteria and viruses, including COVID-19, over a sustained period.
It has been developed in a collaboration between the University of Sydney and University of Melbourne.
Co-lead author University of Sydney’s School of Biomedical Engineering Professor Antonio Tricoli and Director of the University of Melbourne’s Graeme Clark Institute, Professor David Nisbet said the spread of viral and bacterial pathogens through contact with surfaces is a leading cause of infection worldwide.
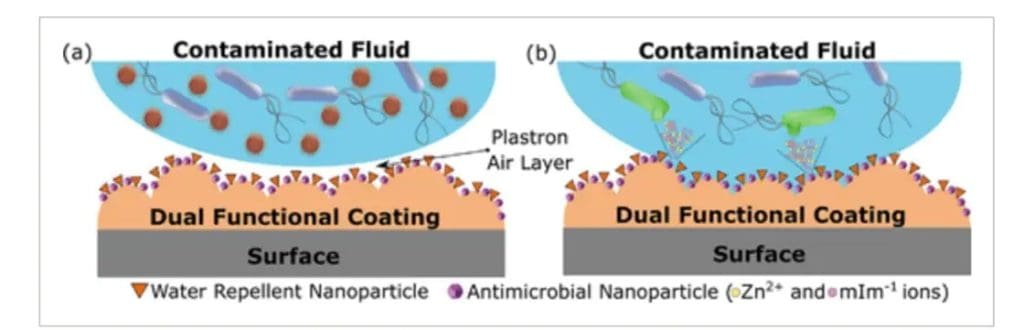
The University of Sydney said an Advanced Science journal article described the spray as being able to repel viruses and bacteria through an air-filled barrier, and kill pathogens through microscopic materials if the layer becomes damaged or submerged for extended periods.
The coating provides a reliable alternative to standard disinfectants, which are becoming less effective and require regular reapplication, and is the only permanent surface layer proven to protect surfaces from contamination by viruses. It is safer than existing alternatives to disinfectant, with no harmful side effects and more stable potency – unlike the next most promising non-disinfectant agent that kills bacteria, silver nanoparticles. The spray uses a combination of plastics strong enough to be considered an alternative to bullet-proof glass, the university said.
Professor Antonio Tricoli said the spray could prevent the surface spread of Foot and Mouth Disease virus from shoes, clothes and travel bags to other surfaces.
“Yes, it would work with the same surface shielding mechanism that we have discovered.
“Basically, the coating would avoid/repeal the adsorption of Foot and Mouth Disease on the surface of those objects, and thus prevent their spreading to other surfaces,” he said.
“It would prevent the uptake of FMD viruses on those surfaces in the first place.”
Professor said retention of the spray’s viral/bacterial surface spread prevention effectiveness would depend on the conditions of usage, and in particular, on how much abrasion is applied to the surface.
“In most cases, it works for about a year or more, but this may be less for high abrasion areas such as the sole of shoes.”
The researchers have established a start-up company to progress the technology and make the spray available commercially, potentially within three years.
The authors said the coating could be applied to surfaces in public settings such as lift buttons, stair rails, surfaces in hospitals, nursing homes, schools and restaurants, to prevent the spread of common viruses and bacteria.
Surface contamination also plays a major role in the evolution of antibiotic-resistant bacterial strains.
“Without a barrier, viruses such as coronaviruses can stay on surfaces and remain infectious for up to a week,” Professor Tricoli said.
“Other viruses such as reoviruses, which can cause colds or diarrhoea, for instance, can remain on surfaces for several weeks, causing large outbreaks in health and aged care facilities.
“Like a lotus leaf, the surface spray creates a coating that repels water. Because the pathogens like to be in water, they remain trapped in the droplets and the surface is protected from contamination,” he said.
“If this mechanism fails, a secondary burst of ions is triggered by carefully designed nanomaterials dispersed in the coating.”
The spray was developed over a five-year collaboration by the multi-university research team and was funded in part by Australian Research Council and NHMRC grants.
The team tested the mechanical stability and surface energy of the coating. They also tested its ability to resist contamination from bacteria and viruses by subjecting it to high concentrations of both. Samples were submerged for extended periods of time and the sprayed surfaces were deliberately damaged to test the spray’s resilience against their contamination.
“We have identified the mechanical processes underpinning how the spray works and quantified its effectiveness in different environments,” Professor Nisbet said.
“For this study, we tested metal surfaces.
“However, in the past we have shown the spray can be applied to any surface, for example, blotting paper, plastic, bricks, tiles, glass and metal,” he said.
“The coating has been engineered through a simple and scalable technique with a careful choice of materials to provide ultra-durability,” Professor Nisbet said.
“We also believe our explanation of the mechanism behind the antimicrobial and antiviral effects could significantly advance research in antipathogen technologies that could see affordable manufacture of an effective surface spray to protect people from viruses and bacteria.”

HAVE YOUR SAY